What is Salt?
In the world of chemistry there are many compounds that are known as ‘salts’, for example, Sodium Chloride (NaCl: Table salt), Magnesium sulfate (MgSO4: Epsom Salt), Calcium Chloride (CaCl2: Used for de-icing), Potassium Nitrate (KNO3: Used in fertilizers), Sodium Acetate (CH3COONa: A common organic salt).
In this article we will be looking primarly at the compound Sodium Chloride – known and referred to as ‘Salt’ – its uses and effects on the human body.
Salt is a naturally occurring substance formed from a combination of the two elements, Sodium (Na+) and
Chloride (Cl-). It exists in nature primarily as the mineral halite and is found in vast quantities in seawater, salt lakes, and underground mineral deposits.
Although chloride is derived from chlorine (Cl2), chloride and chlorine are not the same; they are different forms of the same element with distinct properties.
How does natural Salt occur?
Natural salt occurs primarily through the evaporation of water, which leaves behind sodium and chloride ions that have accumulated from weathered rocks, volcanic activity, and hydrothermal vents. Over millions of years, these processes have created vast underground rock salt (halite) deposits from ancient dried-up seas, as well as salt-rich lakes and oceans.
How Natural Salt Forms
- From Seawater:
Salt does not exist as a solid in the ocean but as dissolved ions (sodium and chloride) in water. When seawater is trapped in shallow ponds or basins and evaporates, due to sun and wind, these ions crystallize into solid salt. - Ancient Seas (Halite or ‘Rock Salt’):
Many underground salt deposits were formed when ancient oceans or lakes were cut off from the main sea and evaporated entirely, leaving thick layers of salt that were later buried by geological activity. - Volcanic & Geological Sources:
Salt ions are introduced into the water cycle through the weathering of terrestrial rocks (carried by rivers) and from seafloor volcanoes and hydrothermal vents.

Photo credt - Ren Fuller
Important Distinction
While salt itself occurs naturally, the table salt found in your kitchen is often refined and processed. This process typically involves purifying the mineral, removing trace minerals, and
sometimes adding iodine or anti-caking agents. However, its fundamental chemical structure (sodium chloride) remains a naturally occurring compound.
Rock salt is coarser, cheaper, and contains fewer chemical anti-caking agents than table salt. While they share the same chemical formula, table salt is refined, whereas rock salt is often
in its raw, natural state.
Fun facts about salt
If you wanted to eat in the winter, you needed salt. If you wanted to sail the oceans, you needed salt to preserve your meat. All your meats and fish, and some other foods,
had to be dried and packed in salt to prevent them from going bad in winter. Salt was critical to life. However, it was labor intensive to produce and transport making it an
expensive commodity.
It was so important that during colonial times, it was called “The Fifth Element” after air, fire, earth and water. And of course, we know that
the word “salary” (salarium), wages or pay, comes from the latin word for salt (sal) when Roman soldiers were paid in salt. The expression,
“He is worth his salt” comes from Greek times when salt was used to buy slaves.
Are Salt and Sodium the same?
No, salt and sodium are not the same thing, though the terms are often used interchangeably in everyday conversation about salt.
The Core Difference
Sodium is a single mineral and a chemical element. It is an essential nutrient that helps your body maintain fluid balance and supports nerve and muscle function.
Salt (scientifically known as sodium chloride) is a chemical compound made of two minerals: sodium (40%) and chloride (60%).
Why the Distinction Matters
When health organizations or nutrition labels refer to “sodium”, they are talking about the specific mineral that can impact blood pressure. While salt is the primary source of sodium in most diets, sodium is also found in other ingredients like baking soda (sodium bicarbonate) and MSG (monosodium glutamate).

Fresh Natural foods are Good healthy foods
What is ‘Correct Information’
When it comes to health It is now becoming more and more challenging for an ordinary person to decide what to believe and what not to believe.
No Peer Reviewed, highly alert, watchful journal is immune to publishing research studies which may later be found bogus or faulty.
Supplements
Supplements may provide some benefit and at time may become essential, but then we should follow the advice of honest medical doctors while thankfully reading the honest scientific medical communities/experts suggestion as explained by Harvard heroes (heroes Scientist / experts) here: Fish-oil, friend or foe?
Read more on Facts on Supplements and Herbs
What is Sodium Chloride and how is it used?
Sodium chloride (NaCl), commonly known as salt, is an ionic compound composed of sodium and chlorine ions. It is crucial for maintaining fluid balance, transmitting nerve impulses, and muscle function in the human body. Beyond dietary use as a seasoning and preservative, it is essential in industrial processes, medical saline solutions, and is used extensively for de-icing roads in winter.
Where does most of the salt in our diet come from?

Most dietary sodium does not come from the salt shaker. In many countries, over 70% to 75% of sodium comes from packaged, processed, and restaurant prepared foods.
Key Aspects of Sodium Chloride:
- Properties:
A white crystalline compound, NaCl is highly soluble in water and has a high melting point of over 800°C due to strong ionic bonds. - Molecular Formula:
The chemical formula represent a 1:1 ratio of sodium (Na+) and chloride (Cl-) ions. - Role in the Body:
It is critical to human health, helping to regulate osmotic pressure, nutrient transport, and proper function of the nervous system and muscles. - Common Uses:
Beyond seasoning, it is used in food preservation, curing, manufacturing chemicals, water softening, and in medical applications like intravenous saline. - Health Effects:
While essential, excessive sodium intake (over 3,400 mg/day) is linked to high blood pressure, heart disease, and stroke.
Common Uses
In cooking and food preparation it is used as a flavor enhancer, food preservative, and ingredient in processed foods. In industry, for example the chemical industry, it is used to produce chlorine and sodium hydroxide, as well as in manufacturing soap, paper, and textiles. Its Medical uses include, Saline solutions (0.9% NaCl) – used to treat the effects of dehydration, intravenous fluids, cleaning wounds, and in eye drops. Other uses Used in de-icing roads in winter, and as a softener for water.
Health Effects & Nutrition
The human body requires a small amount of sodium (~500mg daily or 1/10th (0.1) of a teaspoon) for vital functions, though the typical intake is generally much higher. Too much salt can lead to high blood pressure, stroke, heart disease, and calcium loss.
Biological Importance
It is the primary salt in seawater, and in the extracellular fluid of many organisms where it maintains Electrolyte Balance. The body uses it in Nutrient Transport where it helps in glucose absorption and nutrient transport across cell membranes.
Sodium Chloride as a Food Ingredient
As a food ingredient, salt (sodium chloride) has multiple uses, such as for curing meat, baking, thickening, retaining moisture, enhancing flavor (including the flavor of other ingredients), and as a preservative. Some common food additives — like monosodium glutamate (MSG: C5H8NO4Na), sodium bicarbonate (baking soda: NaHCO3), sodium nitrite (NaNO3) commonly used as a food preservative for cured meats that helps preserve color and prevent bacterial growth, and sodium benzoate (C6H5COONa) a widely used, water-soluble preservative (E211) that prevents mold, bacteria, and fungi growth in acidic foods, beverages, and personal care products — also contain sodium and contribute (in lesser amounts) to the total amount of “sodium” listed on Nutrition Facts labels.
Some foods that don’t taste salty can still be high in sodium, which is why using taste alone is not an accurate way to judge a food’s sodium content. For example, whilst some
foods that are high in sodium (like pickles and soy sauce) taste salty, there are also many foods (like cereals and pastries) that contain sodium but don’t taste salty - possibly because
they may be high in sugars, or flavourings.
Also, some foods that you may eat several times a day (such as breads) can add up to a lot of sodium over the course of a day, even though an individual serving may not be high in sodium.
What is Sodium?
Sodium (Na) is an essential mineral and electrolyte that regulates blood pressure, body fluids, and nerve/muscle function. Adults should limit daily intake to less than 2,300 mg (about one teaspoon of salt), as excessive intake causes high blood pressure, while deficiency – rare in healthy individuals – leads to cell swelling. Reduce intake by checking food labels, minimizing the eating of processed foods, and cooking at home. While the body only requires about 500 mg per day to function, most people exceed the recommended limit of 2,300 mg per day, which can lead to high blood pressure and increased risks of heart disease and stroke.
Uses of Sodium
Sodium has a significant Biological Function where it is essential for maintaining plasma volume, acid-base balance, nerve impulses, and nutrient absorption. In Dietary Usage it is primarily consumed as sodium chloride (table salt) to enhance food flavor and to preserve food. Its Industrial Applications include use in the manufacture of chemical compounds such as sodium carbonate (Na2CO3) used in glass making, detergent production, water softening, and as a cooling agent in nuclear reactors.
Why Your Body Needs Sodium
Health Effects of Sodium

Sodium is an essential nutrient – meaning your body cannot survive without it – that acts as a critical electrolyte to maintain fluid balance, conduct nerve impulses, and support muscle contractions and relaxation. However, excessive Intake brings risks where high intake is linked to high blood pressure (hypertension), cardiovascular disease, and edema. Too little sodium can result is Deficiency Symptoms (Hyponatremia). Though rare in healthy adults, low levels can cause headaches, fatigue, dizziness, muscle cramps, and in severe cases, seizures or coma due to cellular swelling.
Sodium plays several vital roles in keeping your systems running, for example Fluid Balance where it helps distribute water properly inside and outside your cells, which is crucial for maintaining blood volume and pressure. In Nerve and Muscle Function, Sodium allows your brain to communicate with your muscles and organs. It is specifically required for your heart and other muscles to contract and relax effectively. Sodium is essential for transporting substances like glucose into your bloodstream.
Recommended Daily Intake
For adults the Dietary Guidelines for Americans advise limiting intake to less than (<2,000mg sodium) per day. Although you only need a small amount (roughly 500 mg), it is very easy to meet, and exceed, this through a standard diet as it is found naturally in many foods, for example meat, milk, shellfish, and is often added to processed items.
What is Chloride
Chloride is an essential electrolyte and negatively charged ion (Cl- ) derived from chlorine. It is crucial for maintaining fluid balance, blood pressure, pH levels, and proper nerve/muscle function in the body. Primarily consumed as sodium chloride (table salt), it is also vital for producing stomach acid to aid digestion.
Chloride works in conjunction with other electrolytes, such as sodium and potassium, to maintain overall physiological health.
How is the amount of Sodium in foods measured and labelled?
Sodium vs. Salt
Countries such as the United States, China and Mexico use “sodium” as the labeling unit, which aligns with international practices. In contrast, Japan, the European Union, the United Kingdom, and Indonesia use “salt” as the labeling unit. This localized approach is more consumer-friendly, as people are more familiar with the concept of “salt” in describing the salt content of food.
Sodium levels in food are measured using standardized laboratory techniques like Inductively Coupled Plasma (ICP) or titration, and are labeled differently depending on the region: the US uses “sodium” in milligrams (mg), while the EU and others often list “salt” in grams (g).
In the EU and UK, sodium and salt levels are managed through strict labeling requirements and laboratory analysis. Manufacturers must declare salt (not sodium) on nutrition labels per 100g or 100ml. To determine these values, labs use analytical methods like ashing followed by spectrophotometry (AAS or ICP). While consumers see “salt” on packaging, the actual sodium content can be calculated by dividing the salt value by 2.54.
Worldwide Labeling Variations
Labeling regulations vary significantly by jurisdiction:
- United States: Requires the listing of Sodium (mg) per serving on the Nutrition Facts panel.
- European Union: Typically mandates the listing of Salt (g) per 100g/100ml. Note that 1g of sodium is roughly equivalent to 2.5g of salt.
- Ingredients: Regardless of the unit, additives like sodium nitrite (NaNO3: used as a food preservative in meats) or sodium bicarbonate (NaHCO3: used as a raising agent in cooking) must generally be listed in the ingredient declaration.
Many countries are adopting front-of-package warning and advice labels (e.g., “High in Sodium”, “Low Sodium”, “Salt Free”, “Low Salt”, or “No Salt” symbols) to simplify consumer choices. However, the design and wording on these symbols vary widely.
Summary Table
| Feature | United States (FDA) | European Union (EFSA) |
|---|---|---|
| Primary Unit | Sodium (mg) | Salt (g) |
| Calculation Basis | Per serving | Per 100g/100ml |
| Focus | Sodium ion | Total Salt (NaCl) |
Natural Foods Containing Sodium
Many natural foods contain sodium, a vital mineral found in small amounts in plants and animals without any additives. Vegetables including celery, beets, celery root, spinach, leafy greens, and root vegetables often contain natural sodium as do milk, meat, poultry, seafood, and eggs. These foods generally contain much lower sodium levels compared to processed alternatives, as the sodium is part of the food's natural structure.

Why should I reduce my salt intake?
Reducing salt intake is crucial to lower high blood pressure, a leading cause of strokes, heart attacks, and cardiovascular disease. Excessive sodium causes the body to retain water, placing strain on blood vessels and kidneys. Reducing intake helps prevent heart failure, lowers stroke risk, decreases dementia risk, and helps prevent kidney damage and osteoporosis
What is the recommended daily intake of salt?
The World Health Organization (WHO) recommends that adults consume less than 5 grams (approximately 1 teaspoon) of salt per day — or less than 2,000 mg of sodium — to reduce blood pressure and risk of cardiovascular disease.
How can you reduce your sodium intake?
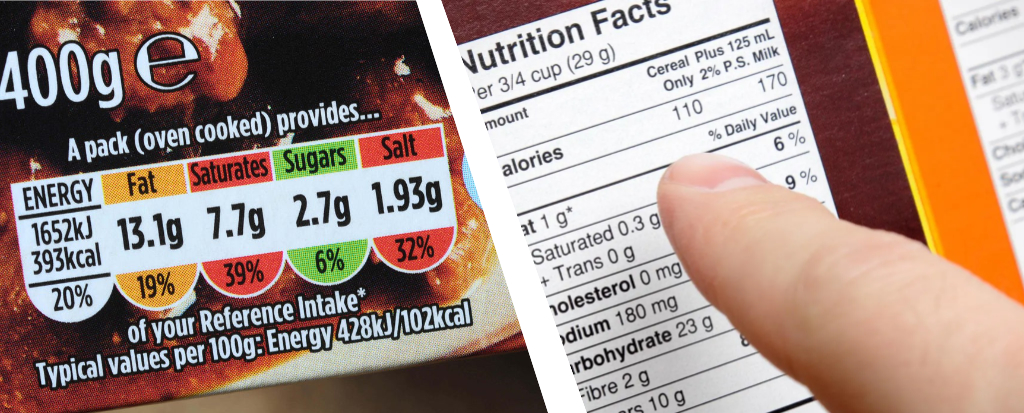
- Read food Nutrition and content Labels:
Check nutrition facts for “Sodium DV” or the Reference Intake (RI) value on food packaging for salt content and choose lower-sodium salt substitutes that contain potassium. - Limit Processed Foods:
Reduce consumption of processed meats, breads, and snacks,limit the use of commercial sauces, dressings and instant products. - Home Cooking:
Prepare meals at home to control salt additions and cook with little or no added sodium/salt; eat mostly fresh, minimally processed foods. - Use Alternatives:
Flavor foods with herbs and spices instead of salt; remove the saltshaker/container from the table.
References and further reading
goqii.com - Interesting facts about Salt you should know
The Nutrition Source - Salt and Sodium
FDA - Sodium in Your Diet - 5 Mar 2024
World Health Organization (WHO) - Sodium reduction - 7 Feb 2025
World Health Organization (WHO) - Measuring salt content of food via direct chemical analysis
World Health Organization (WHO) - Healthy Diet
FDA - Sodium on the Nutrition Facts Label
FDA - Sodium Reduction in the Food Supply
World Health Organization (WHO) - Sodium Reduction
LINKS:

